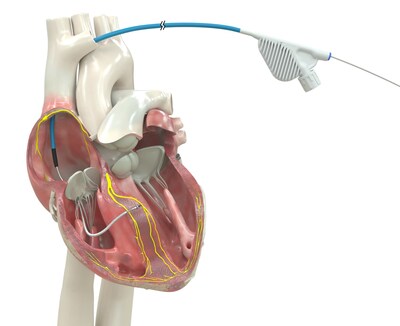
Medtronic OmniaSecure™ defibrillation lead is the first lead of its kind to receive FDA approval for conduction system pacing
Rhea-AI Summary
Medtronic (NYSE: MDT) received FDA approval (March 23, 2026) for an expanded indication for the OmniaSecure defibrillation lead to enable placement in the left bundle branch (LBB) area for conduction system pacing (CSP).
The lumenless, 4.7 French lead is approved for adults and adolescent patients ages 12+, showed 100% defibrillation success at implant and a 2.1% OmniaSecure-related major complication rate at three months in the LEADR LBBAP trial, and commercially launched in the U.S. in January 2026.
Positive
- FDA approval for LBB area conduction system pacing (March 23, 2026)
- Defibrillation success at implant: 100% in LEADR LBBAP
- Three-month major complication rate related to OmniaSecure: 2.1%
- Smallest diameter defibrillation lead on market: 4.7 French
- Approved for adolescents ages 12 and up
- U.S. commercial launch of OmniaSecure in January 2026
Negative
- Longer-term outcomes pending: extended LBB area and LOT-CRT results to be presented at HRS 2026
- Not approved for pediatric patients under age 12
News Market Reaction – MDT
On the day this news was published, MDT gained 1.17%, reflecting a mild positive market reaction.
Data tracked by StockTitan Argus on the day of publication.
Key Figures
Market Reality Check
Peers on Argus
MDT was down 0.82% while key peers were mixed: SYK, BSX, ZBH, and PHG declined, but EW gained 0.78%. With peers moving in both directions and no broad momentum signal, trading appears more stock-specific than sector-driven.
Previous Fda approval Reports
| Date | Event | Sentiment | Move | Catalyst |
|---|---|---|---|---|
| 2026-02-02 | Diabetes FDA clearance | Positive | -0.2% | Medicare access and new FDA clearances expanding MiniMed 780G insulin options. |
| 2025-09-19 | Bladder device approval | Positive | -0.5% | FDA approval of Altaviva ITNM device for urge urinary incontinence treatment. |
| 2025-09-02 | 780G system expansion | Positive | +0.6% | FDA milestones integrating 780G with Instinct sensor and type 2 diabetes use. |
| 2025-04-18 | Sensor FDA approval | Positive | -1.0% | FDA approval of Simplera Sync sensor for use with MiniMed 780G system. |
| 2025-02-24 | DBS system approval | Positive | +0.3% | FDA approval of BrainSense Adaptive DBS and Electrode Identifier for Parkinson’s. |
Recent FDA approvals for MDT have produced small, mixed next-day moves, averaging about -0.18%, with slightly more divergences than alignments.
Over the past year, Medtronic has repeatedly announced U.S. FDA approvals across diabetes, bladder control, and neuromodulation. These milestones, such as new sensors, expanded MiniMed 780G indications, and the BrainSense Adaptive DBS system, broadened its portfolio but historically led to modest share-price reactions, averaging -0.18% the next day. Today’s OmniaSecure lead approval for conduction system pacing adds another regulatory win, this time in cardiac rhythm management, extending the pattern of frequent approvals with relatively muted immediate stock responses.
Historical Comparison
In the past year, MDT logged 5 FDA-approval headlines with an average next-day move of -0.18%, suggesting that even meaningful regulatory wins have historically produced only modest price reactions.
Recent FDA approvals span diabetes tech, bladder neuromodulation, and adaptive deep brain stimulation. The OmniaSecure lead approval extends this regulatory cadence into cardiac rhythm management, broadening Medtronic’s portfolio of advanced, sensor-driven and physiologic therapies across multiple therapeutic areas.
Regulatory & Risk Context
An effective S-3ASR shelf filed on 2026-03-09 allows Medtronic and affiliated issuers to offer debt securities from time to time, fully and unconditionally guaranteed on a joint and several basis by designated group members. Specific amounts and terms will be set in future prospectus supplements.
Market Pulse Summary
This announcement adds a notable FDA approval to Medtronic’s cardiovascular portfolio, making OmniaSecure the first defibrillation lead cleared for conduction system pacing in the left bundle branch area, with 100% implant defibrillation success and a 2.1% major complication rate. In context of multiple recent FDA approvals across other franchises, investors may focus on clinical uptake, real-world performance beyond the LEADR trial, and how this innovation contributes to broader cardiac rhythm management growth.
Key Terms
conduction system pacing medical
left bundle branch medical
cardiac resynchronization therapy medical
implantable cardioverter-defibrillator medical
ventricular tachyarrhythmias medical
ventricular fibrillation medical
lumenless technical
extravascular medical
AI-generated analysis. Not financial advice.
Built for high reliability, world's only lumenless defibrillation lead approved for placement in the left bundle branch area
GALWAY,
The OmniaSecure defibrillation lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat potentially life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. In addition to being the first defibrillation lead approved for placement in the LBB area, the OmniaSecure lead is the smallest diameter defibrillation lead on the market (4.7 French, or 1.66 mm) and the only one approved for adults and adolescent pediatric patients ages 12 and up.
Engineered based on the reliable Medtronic SelectSecure™ Model 3830 pacing lead − the first pacing lead to receive FDA approval for CSP in 2022 − the OmniaSecure defibrillation lead is built for high reliability and durability with its lumenless construction (no hollow channel on the inside), which enables the lead to be delivered via catheter for precise placement in the right ventricle, in addition to the LBB area. Conduction system pacing works by tapping into the heart's natural electrical system, providing patients needed therapy while avoiding cardiomyopathy or other complications sometimes associated with traditional pacing methods.1
"Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart's natural electrical system to enable a more synchronous, physiologic pattern," said Trevor Cook, vice president and general manager of the Defibrillation Solutions business, which is part of the Cardiac Rhythm Management operating unit at Medtronic. "This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients."
ICDs and CRT-Ds are the gold standard for preventing sudden cardiac arrest. For patients who require chronic pacing and defibrillation therapy, the OmniaSecure lead can offer treatment for dangerously fast arrhythmias while delivering conduction system pacing through the LBB area, when connected to a transvenous ICD or CRT-D, such as the Medtronic Cobalt™ and Crome™ family of devices. Patients indicated for CRT may benefit when OmniaSecure is paired with a left ventricular coronary sinus lead (i.e., biventricular or LOT-CRT configurations). For patients who do not require chronic pacing, the Medtronic extravascular Aurora EV-ICD™ system offers protection as the only ICD with a defibrillation lead positioned outside the vascular space that also provides anti-tachycardia pacing (ATP) in a single device.
FDA approval for the OmniaSecure defibrillation lead was supported by data from the global LEADR LBBAP trial (Lead Evaluation for Defibrillation and Reliability in Left Bundle Branch Area Pacing), which demonstrated the lead's safety and effectiveness. Late-breaking data from the study presented at Heart Rhythm 2025, and later published in Heart Rhythm, showed that the lead delivered high defibrillation success at implant (
"The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success," said Pugazhendhi Vijayaraman, M.D., cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center in
The approval expands the comprehensive Medtronic portfolio of lumenless leads and accessories approved by FDA that enable conduction system pacing. This includes the SelectSecure Model 3830 pacing lead, now implanted in more than one million patients globally, as well as the recently cleared C320LBB delivery catheter and the 5944RL rotatable connector. Together, these CSP solutions deliver stability and long-term performance that further enhance procedural precision and versatility.
Following FDA approval for placement in traditional locations in the right ventricle, the OmniaSecure defibrillation lead launched commercially in the
About Medtronic
Bold thinking. Bolder actions. We are Medtronic. Medtronic plc, headquartered in Galway,
Any forward-looking statements are subject to risks and uncertainties such as those described in Medtronic's periodic reports on file with the Securities and Exchange Commission. Actual results may differ materially from anticipated results.
Contacts:
Joey Lomicky
Public Relations
+1-763-526-2494
Ingrid Goldberg
Investor Relations
+1-763-505-2696
1 Padala SK and Ellenbogen KA. Card Electrophysiol Clin. 2021;13(4):755-84. |
2 Vijayaraman P, et al. Heart Rhythm. Published online October 17, 2025. |
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-omniasecure-defibrillation-lead-is-the-first-lead-of-its-kind-to-receive-fda-approval-for-conduction-system-pacing-302720215.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/medtronic-omniasecure-defibrillation-lead-is-the-first-lead-of-its-kind-to-receive-fda-approval-for-conduction-system-pacing-302720215.html
SOURCE Medtronic plc
FAQ
What FDA approval did Medtronic (MDT) announce for the OmniaSecure lead on March 23, 2026?
What clinical results supported Medtronic's OmniaSecure FDA approval (MDT)?
Which patients are eligible for the OmniaSecure defibrillation lead (MDT) after the approval?
What are the device features of Medtronic's OmniaSecure lead (MDT) relevant to clinicians?
When did Medtronic (MDT) begin commercial sales of the OmniaSecure defibrillation lead in the U.S.?